Gold nanostars-based SERS nanotags for bioimaging
American scientists published in Nanotheranostics a comprehensive review about design concepts for SERS nanotags with a particular emphasis on the development of SERS nanotags for bioimaging and theranostics. Under the term “design parameters,” authors mean tailoring the Raman reporter molecule, surface coating, and targeting moieties for the intended application, as well as fine-tuning the characteristics of the plasmonic core.
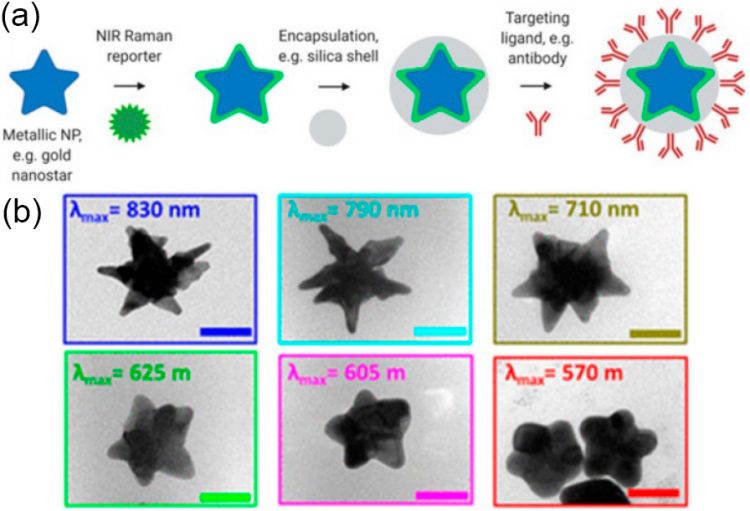
SERS nanoparticles are typically made up of a plasmonic nanoparticle core coated with molecules (Raman reporters) and enclosed in a protective shell that can be covered with targeted ligands for biological purposes (see Figure 1(a)). Raman reporters are chosen for their polarizability, resonance qualities, and binding affinity to the metal NP surface, which multiplies the overall number of reporters per particle, resulting in higher signal intensity. A biocompatible coating encases the metal/molecule system to stabilize the metal nanoparticle and prevent desorption of the Raman reporter molecules from the NP surface. After all, SERS nanoparticles can be decorated with targeting ligands that drive the nanotags toward specific disease indicators, allowing detection of them with Raman imaging. In this work, scientists explain: how the nanoparticle's shape impacts its localized surface plasmon resonance (see Figure 1(b)), which metal is preferable for biological applications, the difference between several synthesis protocols, how to choose Raman reporters and coatings, etc. Authors claim that preclinical testing of gold nanostar-based nanoparticles has demonstrated their enormous potential for disease (marker) detection and even therapeutic intervention via photothermal therapy.
Figure 1. Schematic representation of SERRS nanotag structure for in vivo imaging applications. (a) Metallic substrates, e.g. gold nanostars, are functionalized with a NIR Raman reporter which are then encapsulated in a biocompatible coating such as silica. Active targeting can be achieved through conjugation of specific targeting moieties, e.g. antibodies to the nanoparticle surface. (b) TEM images of gold nanostars with different localized surface plasmon resonance maxima. Adapted with permission from Plasmon Tunability of Gold Nanostars at the Tip Apexes. Adopted from here.